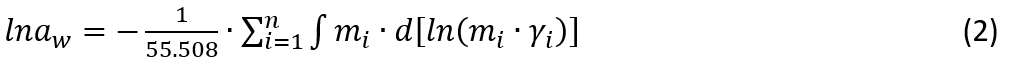Difference between revisions of "Comparing activity coefficients between different thermodynamic frameworks"
| Line 22: | Line 22: | ||
[[File:Eqn2.png]] | [[File:Eqn2.png]] | ||
| − | Only binary solution (e.g. NaNO<sub>3</sub>+H<sub>2</sub>O) is considered here to simplify the explanation. For an electrolyte that is fully dissociated in the aqueous solution (e.g. NaNO<sub>3</sub> in the MSE model), the molality of Na<sup>+</sup> and NO<sub>3</sub><sup>-</sup> are the same as the total NaNO<sub>3</sub> concentration. If ion-pairing is considered (e.g. NaNO<sub>3</sub> in the AQ model), the Na<sup>+</sup> and NO<sub>3</sub><sup>-</sup> concentrations will be different from (i.e. smaller than) the total NaNO<sub>3</sub> concentration due to the formation of ion-pair, NaNO<sub>3(aq)</sub>. In this case, the molality (m) and activity coefficient (γ) of NaNO<sub>3(aq)</sub> will also need to be included in Eq. (1) and (2). The value of activity coefficient for each species will be determined based on concentrations of all species present (i.e. Na+ and NO3- in the MSE mode, and Na<sup>+</sup>, NO<sub>3</sub><sup>-</sup>, NaNO<sub>3(aq)</sub> in the AQ model, in addition to other less important species such as H<sup>+</sup>/H<sub>3<sub>O<sup>+</sup> and OH<sup>-</sup>). | + | Only binary solution (e.g. NaNO<sub>3</sub>+H<sub>2</sub>O) is considered here to simplify the explanation. For an electrolyte that is fully dissociated in the aqueous solution (e.g. NaNO<sub>3</sub> in the MSE model), the molality of Na<sup>+</sup> and NO<sub>3</sub><sup>-</sup> are the same as the total NaNO<sub>3</sub> concentration. If ion-pairing is considered (e.g. NaNO<sub>3</sub> in the AQ model), the Na<sup>+</sup> and NO<sub>3</sub><sup>-</sup> concentrations will be different from (i.e. smaller than) the total NaNO<sub>3</sub> concentration due to the formation of ion-pair, NaNO<sub>3(aq)</sub>. In this case, the molality (m) and activity coefficient (γ) of NaNO<sub>3(aq)</sub> will also need to be included in Eq. (1) and (2). The value of activity coefficient for each species will be determined based on concentrations of all species present (i.e. Na+ and NO3- in the MSE mode, and Na<sup>+</sup>, NO<sub>3</sub><sup>-</sup>, NaNO<sub>3(aq)</sub> in the AQ model, in addition to other less important species such as H<sup>+</sup>/H<sub>3</sub>O<sup>+</sup> and OH<sup>-</sup>). |
Revision as of 14:21, 28 September 2020
Question:
I am comparing the activity coefficient of the nitrate ion (NO31-). I looked up the value in OLI Studio for the AQ thermodynamic framework and also in the MSE thermodynamic framework. They differ by over 50% with the MSE derived values being much larger. Why?
Response: From Peiming Wang, PhD. OLI Systems
I agree with the concept that obtaining different activity coefficient values from the two different models (i.e. AQ vs. MSE) under the same total salt concentration, temperature, and pressure conditions if speciation is different (e.g. with or without the ion-pair such as NaNO3(aq) in the NaNO3 solution). This is because activity coefficients are a function of each and all individual species (ions or neutral), and the speciation in the two models are different, resulting in different equilibrium concentrations for individual species, and thus, different activity coefficients.
Below, I am hoping to provide further reasoning on the basis of thermodynamic fundamentals.
Part 1.
In an equilibrium solution at a given temperature and pressure, Gibbs-Duhem equation must hold, i.e.,
where ai is activity of species i: ai=mi∙γi and a0=aw (water activity), and n is the number of species in the solution. Equation (1) can be integrated as (where n0 is set to be 55.508 for 1 kg H2O):
Only binary solution (e.g. NaNO3+H2O) is considered here to simplify the explanation. For an electrolyte that is fully dissociated in the aqueous solution (e.g. NaNO3 in the MSE model), the molality of Na+ and NO3- are the same as the total NaNO3 concentration. If ion-pairing is considered (e.g. NaNO3 in the AQ model), the Na+ and NO3- concentrations will be different from (i.e. smaller than) the total NaNO3 concentration due to the formation of ion-pair, NaNO3(aq). In this case, the molality (m) and activity coefficient (γ) of NaNO3(aq) will also need to be included in Eq. (1) and (2). The value of activity coefficient for each species will be determined based on concentrations of all species present (i.e. Na+ and NO3- in the MSE mode, and Na+, NO3-, NaNO3(aq) in the AQ model, in addition to other less important species such as H+/H3O+ and OH-).