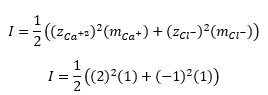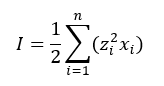Difference between revisions of "Ionic Strength"
(→Ionic Strength (molal based or m-based)) |
|||
| Line 3: | Line 3: | ||
The ionic strength is a quantity representing the strength of the electric field in a solution, and it is equal to the sum of the molalities of each type of ion present multiplied by the square of their charges, as represented by the following equation: | The ionic strength is a quantity representing the strength of the electric field in a solution, and it is equal to the sum of the molalities of each type of ion present multiplied by the square of their charges, as represented by the following equation: | ||
| − | [[File: ionic molal | + | [[File: ionic molal based1.png ]] |
Where n is the number of charged species. | Where n is the number of charged species. | ||
| Line 11: | Line 11: | ||
| − | [[File:molal | + | [[File:molal based2.png]] |
Revision as of 07:21, 21 July 2020
Ionic Strength (molal based or m-based)
The ionic strength is a quantity representing the strength of the electric field in a solution, and it is equal to the sum of the molalities of each type of ion present multiplied by the square of their charges, as represented by the following equation:
Where n is the number of charged species.
For example, a 1.0 molar solution of NaCl has 1.0 moles of Na^+ions and 1.0 moles of Cl^- ions in 1 kg of H2O. Therefore, the ionic strength is 1.0 molal.
Now, consider a 1.0 molal solution of CaCl2. This solution has 1.0 moles of Ca^(+2) ions and 2.0 moles of Cl^- ions in 1 kg of H2O. Therefore, the ionic strength is 3.0 molar, or it can be said that a 1.0 molal solution of CaCl2 behaves similar to a 3.0 molar solution of NaCl.
Ionic Strength (mole fraction based or x-based)
In this case the ionic strength is calculated using the mole fraction rather than the molality:
Where n is the number of charged species.