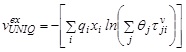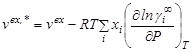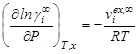Difference between revisions of "MSE Density Parameter Update"
| Line 24: | Line 24: | ||
| − | |||
[[File:T22.jpg]] [[File:T23.jpg]] (Correspond to D0JI, D1JI, D2JI) | [[File:T22.jpg]] [[File:T23.jpg]] (Correspond to D0JI, D1JI, D2JI) | ||
Equation (3) is equivalent to the equation: | Equation (3) is equivalent to the equation: | ||
| Line 35: | Line 34: | ||
Here [[File:T8.jpg]] is calculated from contributions of UNIQUAC and “middle-range” terms, if applicable, and [[File:T27.jpg]] is from either HKF or as described in 2002 paper for organic component. | Here [[File:T8.jpg]] is calculated from contributions of UNIQUAC and “middle-range” terms, if applicable, and [[File:T27.jpg]] is from either HKF or as described in 2002 paper for organic component. | ||
| + | |||
| + | [[Category:Tips]] [[Category:Thermodynamics]] | ||
Revision as of 07:36, 11 July 2016
In UNIQUAC term, the excess molar volume,![]() , is assumed to be analogous to that of the excess Gibbs energy, i.e.
, is assumed to be analogous to that of the excess Gibbs energy, i.e.
where ![]() is a different interaction parameter from that of
is a different interaction parameter from that of ![]() in the
in the ![]() model.
The symmetrically normalized
model.
The symmetrically normalized ![]() (subscript UNIQ is omitted in the following text) needs to be converted to the unsymmetrically normalized value (
(subscript UNIQ is omitted in the following text) needs to be converted to the unsymmetrically normalized value (![]() ) for density calculations in MSE.
Conversion from
) for density calculations in MSE.
Conversion from![]() to
to ![]() :
Standard state chemical potentials in the two normalizations are related by
:
Standard state chemical potentials in the two normalizations are related by
![]() (or
(or ![]() )
which is derived from equality of chemical potentials in two normalizations, and using
)
which is derived from equality of chemical potentials in two normalizations, and using ![]() 1 in the infinite dilution limit. Derivatives of ’s with respect to pressure give volumes, i.e.
1 in the infinite dilution limit. Derivatives of ’s with respect to pressure give volumes, i.e.
![]() ->
-> ![]() Here is how to calculate
Here is how to calculate ![]() Analogous to
Analogous to ![]() ,
, ![]() can be defined as
can be defined as 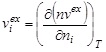 and derived from Eq. (1) as:
and derived from Eq. (1) as:
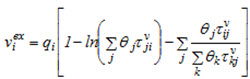 (2a)
(2a)
Now,![]() can be calculated from Eq. (3) by combining Eq (1) and (2b):
can be calculated from Eq. (3) by combining Eq (1) and (2b):
In Eq. (1) and (2),
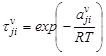
![]() (Correspond to D0JI, D1JI, D2JI)
Equation (3) is equivalent to the equation:
(Correspond to D0JI, D1JI, D2JI)
Equation (3) is equivalent to the equation:
Solution volume is then calculated by
Here ![]() is calculated from contributions of UNIQUAC and “middle-range” terms, if applicable, and
is calculated from contributions of UNIQUAC and “middle-range” terms, if applicable, and ![]() is from either HKF or as described in 2002 paper for organic component.
is from either HKF or as described in 2002 paper for organic component.