Modeling the Chemistry of Carbon Dioxide - Rich Phases with Impurities
Dense Phase CO2 Corrosion: Modeling the Chemistry of CO2 – Rich Phases with Impurities
- Objectives:
- - Model the solubility of So in CO2 - rich phases in order to predict whether solid So can drop out in transmission lines
- - Predict whether So can undergo reactions in the presence of water
- Chemical subsystems to be modeled
- - Pure So
- Volatility of pure So provides a baseline for its solubility in gas phase
- - So – H2O
- Solubility of So in H2O is important if an aqueous phase forms
- Model development requires considering the So – H2O system because the reference state for liquid-phase species is infinite dilution in water
- - So – CO2
- Solubility of So in CO2
- - Redox reactions of sulfur in aqueous media
- Will be important for future modeling of reactions of SOx and NOx
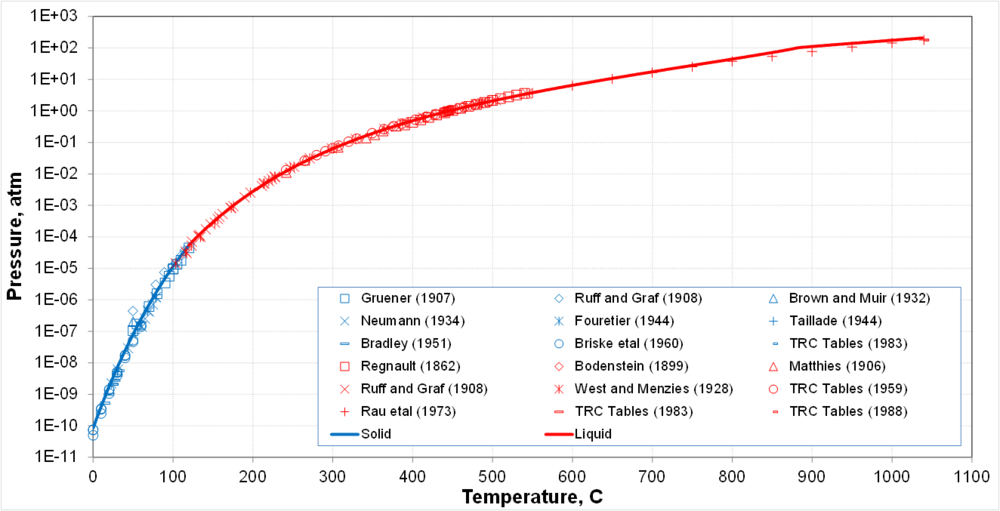
- - Pure So
- Polymeric species of sulfur
- - Numerous sulfur species up to S20 have been detected
- - In the gas phase, eight multimers (So1 through So8) have been assumed in the model
- Thermochemical data are well established for So1 through So8
- So8 is dominant at normal and moderate conditions
- Lower multimers become prevalent at higher temperatures
- - In the pure liquid and solution phase, So8 predominates
- - In the CO2 phase, solvated So-CO2 species may appear
- Solid-vapor equilibrium transitions into liquid-vapor equilibrium at the triple point
- Calculations are supported by a large body of generally consistent data