Creating air headspace
All OLI calculations are closed systems
This means that air is never included unless explicitly created. Below are two images that describe this.
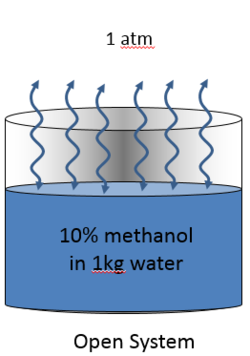
The first image is a 10% methanol in water System open to the Air Environment. The 10% methanol can evaporate and condense. Its mass balance is therefore not conserved. Energy can also transfer to and from the environment. The air mass assumed to be infinite relative to the liquid mass.
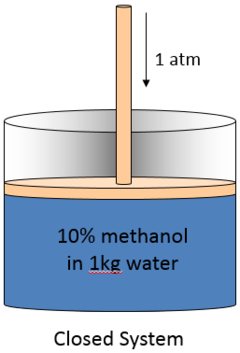
The second image is 10% methanol closed to the environment. Mass cannot leave or enter the system. it is therefore conserved. Heat transfer is allowed.
Creating an Open system
It is impossible to create a true open system using OLI because a total mass is needed for the calculation. However, it is possible to approximate an open system.
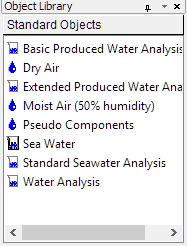
Create an Air stream. Two air streams exists in the Object Library (Menu: View>Toolbars>Object Library). You can drag one of these to the Navigator pane.
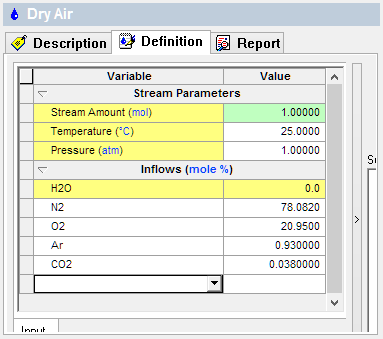
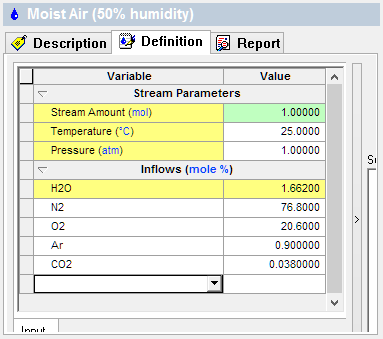
The first image shows the Dry air composition and the second is for Moist air (50% humidity). Notice that the Stream Amounts are 1 mole, or ~24L @25C.
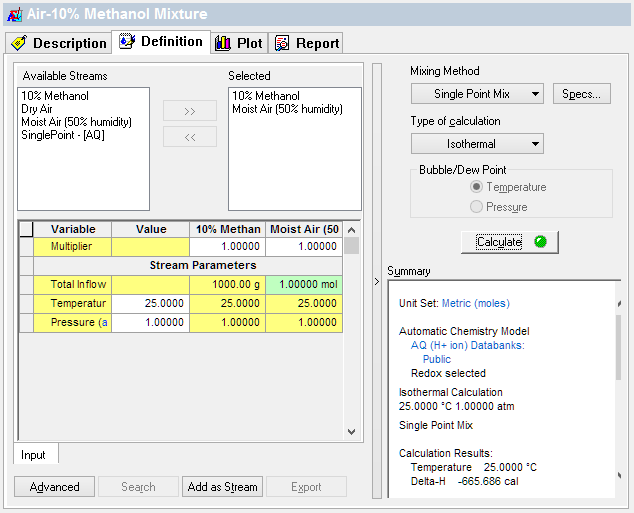
Simulate the open system by combining the Air stream to the Liquid stream in a Mixer. The image below is for a 1kg water in contact with 1mole (24 liters) of air.
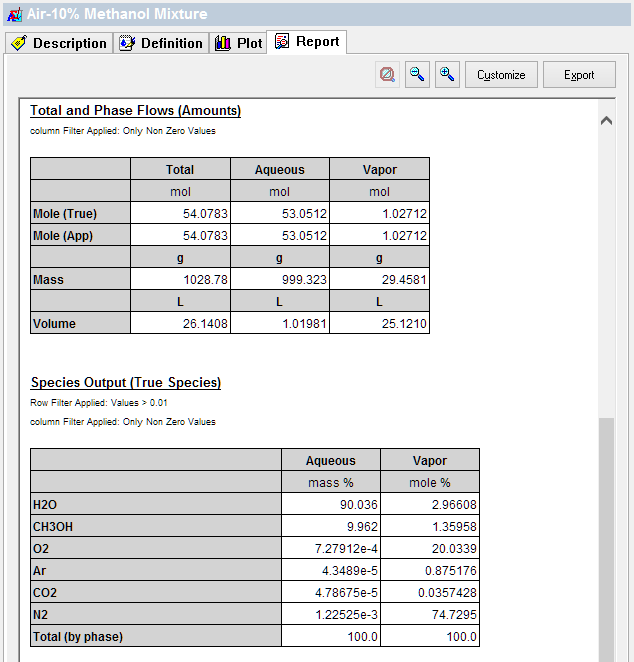
The final mixture is shown below. Note the CO2 concentration (0.036 mole%). This value should be close enough to the original value of 0.038 mole% to ensure that sufficient air is used to simulate an open system.
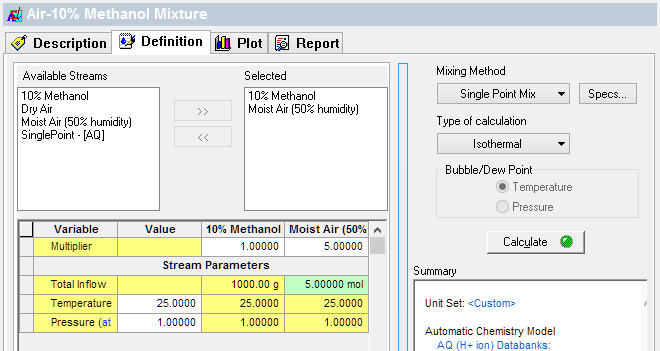
If your calculated CO2 value is too low (e.g., 0.025 mole%) then increase the air amount. Change the Multiplier value in the Definition tab. The image below shows the Air increased to 5 moles (~120 liters).