Difference between revisions of "Scaling Tendencies"
(→What is a Scaling Tendency?) |
(→What is a Scaling Tendency?) |
||
| Line 29: | Line 29: | ||
| − | + | ===Scaling Tendency and Prescaling Tendency in OLI Software=== | |
| − | + | Scaling tendencies are essentially saturation ratios. When the scaling tendency for a species is 1.0, it indicates that this species is in equilibrium with water. In other words, the species is in the solid phase. A scaling tendency below 1.0 indicates sub-saturation and that the solid phase will not form. | |
| − | + | Scaling Tendency is the saturation ratio after all potential solids come to equilibrium. This is the true equilibrium condition (time=∞). | |
| + | Prescaling Tendency represents the condition before any solids are allowed to form. This is a non-equilibrium condition and can be viewed as the condition where time equals 0. | ||
| − | + | For the usage of the term in the software, it is defined as | |
| − | |||
| − | |||
| − | + | Definition for Pre-Scaling tendencies: They are scaling tendencies of a solid before we permit any other solid phases to appear, i.e. before we allow solids to form. This measure acts as if other solids do not exist. The common ion effect is not considered while these tendencies are calculated. | |
| − | |||
| − | |||
| − | |||
Post –Scaling tendencies: Effect of common ions is considered. These tendencies are calculated after solids are formed. Technically the scaling tendencies which you see in the plot in analyzers are these post scaling tendencies. | Post –Scaling tendencies: Effect of common ions is considered. These tendencies are calculated after solids are formed. Technically the scaling tendencies which you see in the plot in analyzers are these post scaling tendencies. | ||
Revision as of 09:14, 29 January 2019
What is Scaling?
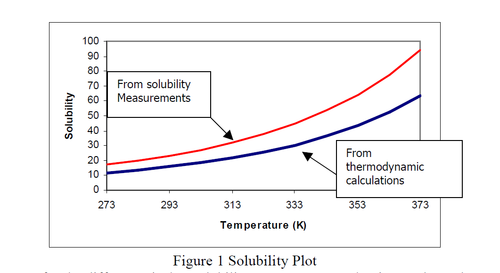
Scaling is the deposition of a mineral salt on processing equipment. Scaling is a result of supersaturation of mineral ions in the process fluid. Through changes in temperature, or solvent evaporation or degasification, the concentration of salts may exceed the saturation, leading to a precipitation of solids (usually crystals).
For example, changes in temperature can cause that calcium bicarbonate precipitates as calcium bicarbonate and releases CO2 into the solution, as is shown in the following reaction:
Ca(HCO3)2(aq) ---> CaCO3(s)+CO2(g)+H2O
The saturation level of a salt in water is a good indicator of the potential for scaling.
What is a Scaling Tendency?
Scaling Tendency is the ratio of the ratio of the concentration of the ions of the salt of interest divided by the equilibrium constant of the same salt (Ksp). For example for the following reaction
CaCO3(s) = Ca+2 + CO3-2
The scaling tendency (ST) is:
ST = [Ca+2][CO3-2]/Ksp
The three possible ST scenarios are the following:
Scaling Tendency Result
< 1.0 The solid is under-saturated and is not expected to form
= 1.0 The solid is saturated and is expected to form
> 1.0 The solid is over-saturated and may or may not form
Scaling Tendency and Prescaling Tendency in OLI Software
Scaling tendencies are essentially saturation ratios. When the scaling tendency for a species is 1.0, it indicates that this species is in equilibrium with water. In other words, the species is in the solid phase. A scaling tendency below 1.0 indicates sub-saturation and that the solid phase will not form.
Scaling Tendency is the saturation ratio after all potential solids come to equilibrium. This is the true equilibrium condition (time=∞).
Prescaling Tendency represents the condition before any solids are allowed to form. This is a non-equilibrium condition and can be viewed as the condition where time equals 0.
For the usage of the term in the software, it is defined as
Definition for Pre-Scaling tendencies: They are scaling tendencies of a solid before we permit any other solid phases to appear, i.e. before we allow solids to form. This measure acts as if other solids do not exist. The common ion effect is not considered while these tendencies are calculated.
Post –Scaling tendencies: Effect of common ions is considered. These tendencies are calculated after solids are formed. Technically the scaling tendencies which you see in the plot in analyzers are these post scaling tendencies.
Pre-scaling tendency is the same formula except for a situation when solids are not allowed to form. The mass transfer between LIQ phase to SOL is not considered while calculating these values. That is the only way to predict 'pre-scaling' tendencies.
For Example, Consider this dissolution
NaHCO3(s) = Na+ + HCO3-
The Ion Activity Product (IAP) is defined as the product of specific ions (in this case the ions resulting from the dissociation of a particular solid).
Let’s consider a 1.0 molal NaHCO3 solution:
IAP = gamma.Na*m.Na*gamma.HCO3*m.HCO3
Assuming Ideal Solution Activities
gamma.Na = 1.0
gamma.HCO3 = 1.0
m.na = 1.0
m.HCO3 = 1.0
IAP = (1.0)(1.0)(1.0)(1.0)
IAP = 1.0
The Solubility Product (KSP) is the thermodynamic limit of ion availability
Ksp = gamma.Na*m.Na*gamma.HCO3*m.HCO3
KSP = 0.403780
The Scaling Tendency is then the ratio of available ions to the thermodynamic limit.
ST = IAP/KSP ST = 1.0/0.403780 ST = 2.48
Was assuming ideal conditions valid??
The actual species concentration and activity coefficients are:
gamma.Na = 0.598
gamma.HCO3 = 0.596
m.na = 0.894
m.HCO3 = 0.866
This results in a different IAP
IAP= (0.598)(0.894)(0.596)(0.866)
IAP=0.276
The new Scaling Tendency is therefore:
ST = IAP/Ksp
ST = 0.276/0.40378
ST = 0.683
Why are the concentrations not equal to 1.0? Speciation and chemical equilibria tend to form complexes which provide a “Sink” for carbonate species. In this example:
CO2o = 0.016 molal
NaHCO3o = 0.101 molal
CO32- = 0.012 molal
NaCO3- = 0.004 molal
What does the Scaling Tendency Mean?
If ST < 1, then the solid is under-saturated If ST > 1, then the solid is super-saturated If ST = 1, then the solid is at saturation Scaling Index = Log (ST)
The post-scaling tendency can go over 1.0 in special cases. If you have disabled a solid from forming (via the Menu Item: Chemistry: Model Options: Phases Tab) then it is possible to have a scaling tendency greater than 1.0. Also if the temperature is above a solid’s Temperature Range (TRANGE) then it can also be above 1.0. Please refer to the TRANGE support tip.
This was OLITips39.