Difference between revisions of "Reactor block in ESP"
| Line 2: | Line 2: | ||
| − | == | + | == Using the reactor block to simulate oxridation/reduction reactions== |
Sometimes it is desirable to simulate oxidation and reduction processes without using the full OLI/Redox chemistry. The advantages are that the reaction is easy to write and takes place irreversibly. The disadvantage is that the reaction must be in terms of molecular inflow species and no oxidation and reduction potential (ORP) will be calculated. | Sometimes it is desirable to simulate oxidation and reduction processes without using the full OLI/Redox chemistry. The advantages are that the reaction is easy to write and takes place irreversibly. The disadvantage is that the reaction must be in terms of molecular inflow species and no oxidation and reduction potential (ORP) will be calculated. | ||
Revision as of 09:16, 30 June 2014
Using the reactor block to simulate oxridation/reduction reactions
Sometimes it is desirable to simulate oxidation and reduction processes without using the full OLI/Redox chemistry. The advantages are that the reaction is easy to write and takes place irreversibly. The disadvantage is that the reaction must be in terms of molecular inflow species and no oxidation and reduction potential (ORP) will be calculated.
In this example, a user wants to oxidize uranium in the +4 oxidation state to the +6 oxidation state using oxygen. The first step is to write the overall reaction that is desired in molecular form. Each component in the reaction must exist in either an OLI databank or in a private databank.
An example of the reaction is this:
Each of these species exist in the OLI databanks as:
Component OLI Name U(OH)4 UIVOH4 O2 O2 UO2(OH)2 UO2OH2 H2O H2O
The equation to be entered into the OLI Reactor block would be:
2UIVOH4 + O2 = 2UO2OH2 + 2H2O
We will now show you the pertinent screens for creating the reactor block.
1. Create a standard OLI Chemistry model
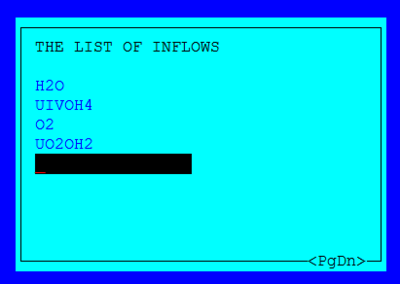
Figure 1 The list of inflows
You are required to include each component in the reaction as a member of the inflow list.
Complete the creation of the chemistry model. Of course, include any other components you require.
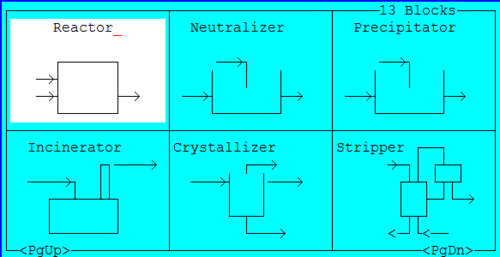
2. Create the reactor block from ESP/Build
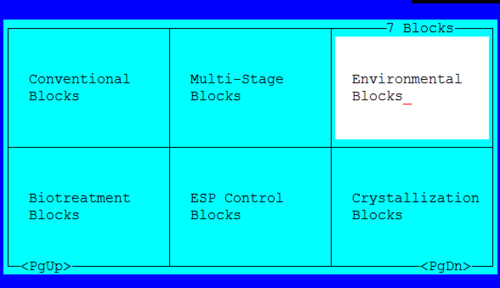
The reactor block is part of the Environmental blocks.
Figure 2 The block layout
Select the Reactor block from the choices.
Figure 3 The Environmental blocks
Select the Aqueous reactor
Figure 4 Selecting the aqueous reactor
Figure 5 the reactor block
Create the input stream(s) and output stream as with any OLI/ESP block.
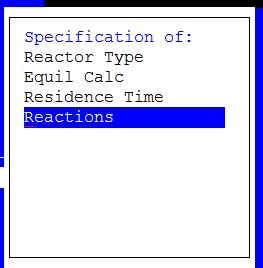
3. Select Parameters from the Action Line
Figure 6 Select reactions
Select the reactions line.
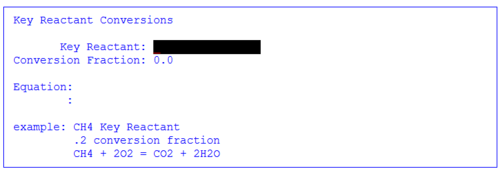
4. Enter the New Reactant
Figure 7 Select new reactant
5. Enter the Key reactant and the equation.
Figure 8 Entering the reaction information
The key reactant is analogous to the limiting reagent. The conversion fraction is the amount of material to be converted. The equation is the reaction.
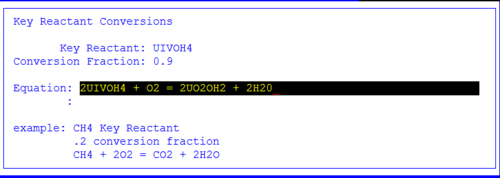
Figure 9 the entered information
In this example, oxygen is oxidizing the +4 UIVOH4 species. The limiting reagent (Key) is specified as UIVOH4. 90 % of the available U+4 will be reacted according to this equation.
There is no time specification for the reactor. The user is required to know the conversion per time.
Use the <End> key to save this information.
Exit the block and run the case.