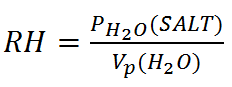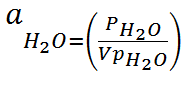Difference between revisions of "Relative Humidity"
(Created page with "The question "What is Relative Humidity?" has multiple answers. The one that makes most sense is "How much water vapor can a solution hold above a salt solution compared to ...") |
|||
| Line 16: | Line 16: | ||
We already calculate the activity of water and report it in the output. Thus if the water activity is 0.95 then we have a relative humidity of 95%. | We already calculate the activity of water and report it in the output. Thus if the water activity is 0.95 then we have a relative humidity of 95%. | ||
| + | |||
| + | Please note, this definition only holds true if you have an aqueous liquid phase (liquid 1) present. If your system is primarily hydrocarbon then this probably an overestimation. | ||
[[user:jberthold | Author: Jim Berthold (OLI)]] | [[user:jberthold | Author: Jim Berthold (OLI)]] | ||
[[Category: Thermodynamics]] | [[Category: Thermodynamics]] | ||
Latest revision as of 09:58, 14 November 2019
The question "What is Relative Humidity?" has multiple answers.
The one that makes most sense is "How much water vapor can a solution hold above a salt solution compared to a pure water system at saturation pressure or temperature?"
This is really a function of the partial pressure of water (PH2O) above a salt solution compared to the pure component vapor pressure of water at the same temperature (T) and pressure (P).
So Relative Humidity is:
Where PH2O(SALT) is the partial pressure of water above the salt solution and Vp(H2O) is the vapor pressure of water.
It turns out that this ratio is also a definition of the activity of water:
We already calculate the activity of water and report it in the output. Thus if the water activity is 0.95 then we have a relative humidity of 95%.
Please note, this definition only holds true if you have an aqueous liquid phase (liquid 1) present. If your system is primarily hydrocarbon then this probably an overestimation.