Autoclave Simulation for Hydro-metallurgy
This simulation takes a known amount of a solid metal oxide (ZnO) and reacts it in an acid/ethanol solution. We will discuss the creation of this process.
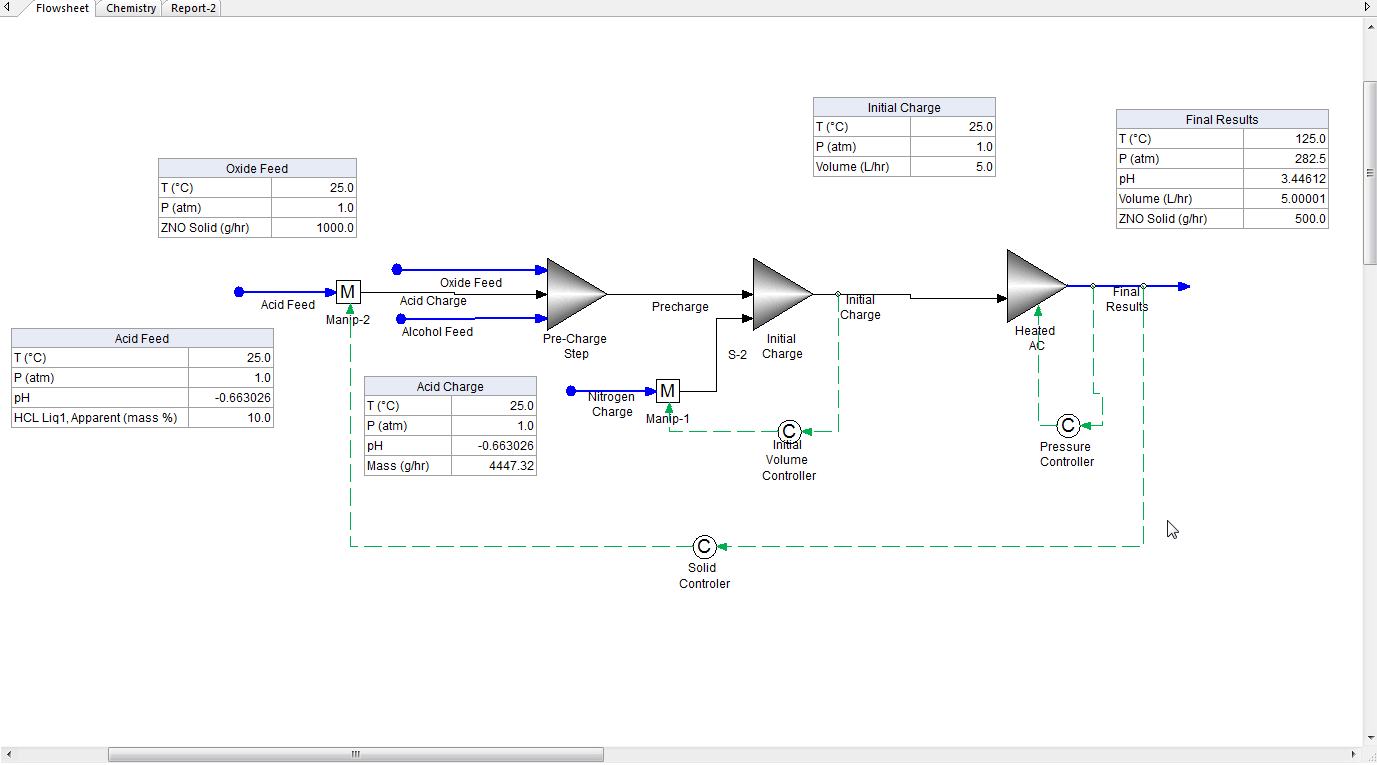
The following image is the completed process. We will not elaborate on the specific steps used to create this process only the overall principles.
The overall objective to determine the final autoclave pressure required to react 1000 grams of zinc oxide (ZnO) in a 10 weight percent solution of hydrochloric acid (HCl). A secondary step is to determine how much initial charge of HCl solution is required to react 50 % (or 500 grams) of the initial metal oxide.
The parameters of the calculation are:
| Specification | Value |
|---|---|
| Autoclave Volume | 5.0 L |
| Initial Temperature | 25.0 C |
| Initial Pressure | 1.0 Atmosperes |
| Final Temperature | 125.0 C |
The chemistry model had the following parameters:
| Specification | Value |
|---|---|
| Thermodynamic Framework | MSE |
| Databanks | Default |
| Inflows | |
| H2O | |
| ZNO | |
| ETHANOL | |
| HCL | |
| N2 |
The simulation of the autoclave involves three steps:
- Load the autoclave with the reactants
- Pressure the autoclave to purge out the head space with an inert gas such as Nitrogen
- Heat the autoclave to the final temperature
We can simulate these three steps using individual unit operations. We have chosen three mixer unit operations.
Step 1: Load the autoclave
- We have created three feed streams
- Oxide Feed
- Acid Feed
- Alcohol Feed
The composition of the feed streams is as follows:
| Stream | Oxide Feed | Acid Feed | Alcohol Feed |
|---|---|---|---|
| Temperature (C) | 25 | 25 | 25 |
| Pressure (Atm) | 1 | 1 | 1 |
| H2O (grams) | 0 | 90 | 0 |
| ZNO (grams) | 1000 | 0 | 0 |
| ETHANOL (grams) | 0 | 0 | 300 |
| HCL (grams) | 0 | 10 | 0 |
| N2 (grams) | 0 | 0 | 0 |
We passed the Acid Feed stream through a manipulate block since we will adjust the flow to match the solid specification of 50% reduction. The three feed streams (and the adjusted acid stream) are sent to a mixer unit operation called Pre-Charge Step
The mixer "Pre-Charge Step" is an isothermal mixer set to a temperature of 25 C and an absolute pressure of 1.0 atmosphere.
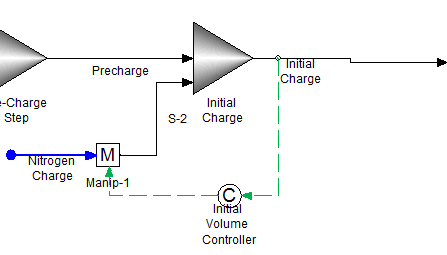
Step 2: Initially charge the autoclave to 5.0 L
This will result in a total volume of less than 5.0 L. We will use nitrogen gas and a feed back controller to adjust the initial charge volume to 5.0 L.
The mixer "Initial Charge" is also an isothermal mixer set to 25 C and an absolute pressure of 1.0 atmospheres. The feedback controller "Initial Volume Controller" is set to adjust the flowrate of the "Nitrogen Charge" stream such that the total volume flow in stream "Initial Charge" is 5.0 L.
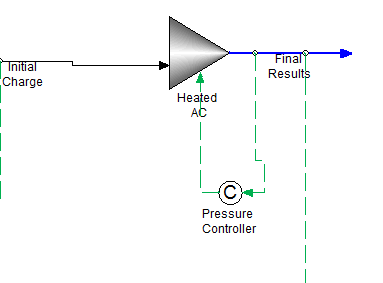
Step 3: Heat the autoclave to the final temperature and determine the final pressure
The effluent of the "Initial Charge" mixer is then sent to the final mixe "Heated AC" Note:the "AC" means Autoclave
This final mixer is also an isothermal mixer set to 125 C and initially 2.0 atmospheres. A feedback controller "Pressure Controller" is set to adjust the mixer pressure to maintain a total volume of 5.0 L
Step 4: How much acid is required to reduce the solid amount to 50% of the original value? What is the final pressure?
This is where adding callouts has tremendous value. In the first image at the top of this page we have the final results. We used one more feed back controller "Solid Controller" to set the final solid mass flow at 500 grams by adjusting the flow of the acid feed through "Manipulate-2".
The initial amount of Zinc Oxide was 1000 grams and in the final result it is 500 grams. We have met the specification of the "Solid Controller." The final pressure is 282.5 atmospheres (~4150 PSIA). The stream "Acid Charge" is the amount of the acid stream needed to met the solid specification. This turned out to be 4.45 Kg/hr.
You can download the worked example file from the following link. This worked example was created in version 9.5.2 of the OLI Flowsheet software. Later versions of the software may have different results.