Calcium Carbonate polymorphs
A question from a user:
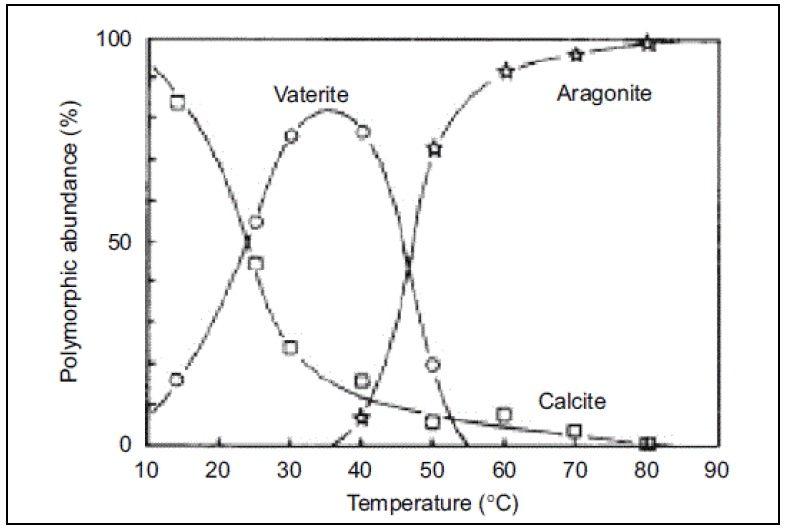
We were just modelling some CaCO3 using the MSE GeoChem (GEMSE) database and MSE Public (MSEPUB). It seems that the it is not predicting the correct polymorph of CaCO3 at high temperatures. i.e., greater than 50 oC. See the graph below.
We never get aragonite to form a solid – doing a simple pH survey with CaCl2 (HCl, NaOH) over a range of temperatures . The information attached is from Scaling of Calcium Carbonate on a heated surface in a flow through system with MEG . Ketil Hyllestad NTNU – Trondheim 2013
Just wanted to know if the Ksp data was correct in the database
A response from Andre Anderko, PhD., FNACE (CTO OLI Systems):
We have investigated the aragonite vs. calcite issue and here is a summary of the findings:
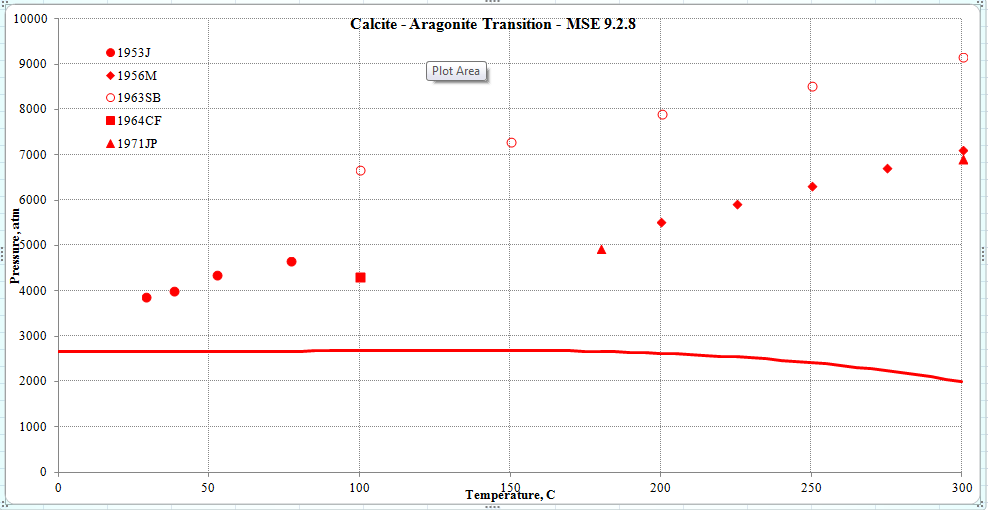
From a thermodynamic point of view, aragonite is a high-pressure form of CaCO3. In the graph below we show the transition pressure above which calcite converts to aragonite.
The line is calculated by MSE and the symbols are experimental. Please note that this is a pure predictions because these data were not used to fit the model (until recently, we were not interested in pressures of 2000 - 9000 atmospheres). MSE predicts the transition qualitatively and underpredicts the data (which are very scattered anyway). In future work we are planning to look more closely into this transition and improve the agreement with the data.
It should be noted that the calcite-aragonite transition happens at practically all temperatures. This means that calcite is always thermodynamically stable at lower pressures. Of course, aragonite can form first as a metastable solid instead of calcite even at low pressures. This happens in nature depending on the environment (especially in many living organisms). However, this cannot be accounted for by thermodynamics alone. The plot of calcite-vaterite-aragonite transformation that was included in the literature plot of Hyllestad must correspond to a kinetically-limited precipitation process.
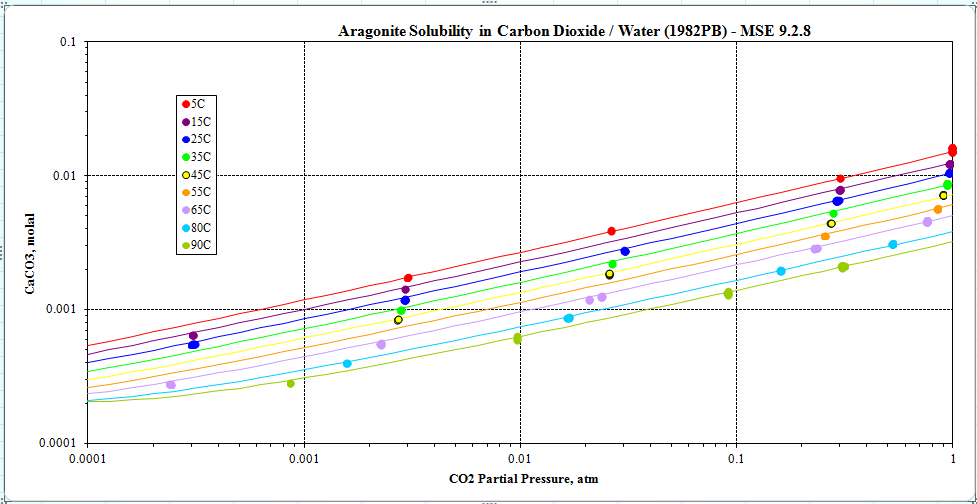
There is no doubt that our Ksp’s for calcite and aragonite are correct because the solubilities of both species are reproduced correctly. The solubility plot for aragonite is shown below :
and we have many similar plots for calcite. To predict the formation of aragonite as a metastable species, it is necessary to remove calcite from the model. Then, the solubility of metastable aragonite should be calculated correctly.