OLI Studio Frequently Asked Questions - FAQ
Frequently asked questions about OLI Studio (including Stream Analyzer, Corrosion Analyzer, Scalechem and EVS)
Contents
- 1 Stream Analyzer
- 1.1 Does OLI Studio have MSE, MSE-SRK and AQ?
- 1.2 Are the Partial Pressures calculated in Autoclaves always at final conditions?
- 1.3 Fixed, point and parent stream conditions - Definitions
- 1.4 Why are my ion concentrations larger in the aqueous phase column than in the total column?
- 1.5 What happened to the Import Database Button in OLI Studio V10
- 1.6 The differences between Standard Volume, Standard Liquid Volume, and Volume?
- 1.7 How to enable the optional calculations in OLI Studio?
- 1.8 How to obtain equilibrium constants in OLI Studio
- 2 Corrosion Analyzer
- 3 ScaleChem
Stream Analyzer
Does OLI Studio have MSE, MSE-SRK and AQ?
MSE and AQ are the heart of our software, they are different models we choose to work with depending on the application we want to model.
Are the Partial Pressures calculated in Autoclaves always at final conditions?
The autoclave calculation was designed to allow the user to specify the partial pressures of CO2, H2S and CH4. The program can then calculate the conditions necessary to achieve those partial pressures.
A common problem in a real-world autoclave is that the partial pressures are known but it is unknown how to charge the autoclave to obtain the desired conditions. Thus the autoclave calculation has ambient and final set of conditions. The ambient is how to charge the autoclave and the final is at operating conditions.
To answer the question, the partial pressures specified in the calculation are always at the conditions specified, either ambient or final conditions.
Fixed, point and parent stream conditions - Definitions
Why are my ion concentrations larger in the aqueous phase column than in the total column?
What happened to the Import Database Button in OLI Studio V10
The differences between Standard Volume, Standard Liquid Volume, and Volume?
How to enable the optional calculations in OLI Studio?
How to obtain equilibrium constants in OLI Studio
Corrosion Analyzer
Corrosion in emulsions
An OLI user recently asked:
Why are we seeing corrosion in the interface liquid (emulsion of HC oil phase) rather than the water phase in our client’s system? What could be corrosive in the emulsion HC phase? This is a water and crude oil solution, and there is a corrosive interface phase.
Andre Anderko (OLI's CTO) replied:
Such phenomena appear from time to time, but they are not always easy to interpret.
The common reason for enhanced corrosion at the interface is overwhelmingly mass transport. A classical example is enhanced corrosion at the seawater/air interface, sometimes referred to as the splash zone. The reason is the enhanced availability of oxygen at the splash zone due to the constant movement in the splash zone and the proximity of the gas phase. If we go deeper below the splash zone, the mass transfer conditions become less favorable to corrosion because the movement of the liquid subsides and oxygen can be transported only by diffusion in a near-stagnant medium.
The situation at the aqueous phase – hydrocarbon emulsion interface is probably qualitatively similar. However, there is a significant difference – the corrosive agent is probably not oxygen, but something else. It is possible that the corrosive agents are the common corrosive components such as CO2 and H2S, but this would require an experimental analysis.
Of course, we cannot model the complex mass transfer phenomena in the splash zone or the two-liquid counterpart of the splash zone. Nevertheless, OLI can model the effect of the corrosive components and we can parametrically increase mass transfer to see if enhanced mass transfer can be responsible for enhanced corrosion.
Corrosion Inhibitors
Question: My research interest is corrosion inhibitor. I will love to know how to optimized corrosion inhibitors in sweet and sour systems in order to reduce cost of testings in the lab. Our research involves the evaluation of corrosion inhibitors for Sweet and Sour condition in the presence of brine at high temperature and high pressure autoclaves. So is your software applicable for our work? How can we use the software in corrosion inhibitor test evaluation and optimization and in predicting the corrosion rates in sweet and sour environments in the absence and present of corrosion inhibitors?
Answer: We have not done any work in the area of corrosion inhibitors as this client is interested in. We have only limited capability to predict the effect of oxyanions on the repassivation potential of specific alloys. But in the absence of corrosion inhibitors, our tool is capable of predicting the corrosion properties such as corrosion rates, corrosion potential, and the repassivation potential for general and localized corrosion of various alloys as a function of operational parameters such as pH, temperature, pressure, fluid compositions and partial pressure of CO2 and H2S
Do you model atmospheric corrosion?
Not directly. If the material from the air condenses then we can simulate the corrosion in drops that condense.
Effects of CO2 corrosion in pipelines
Question: Do you model the effect of contamination in transport supercritical CO2 by pipeline. It was part of an evaluation exercise for carbon sequestration. It indicate that in some circumstances any aqueous phase would be very corrosive.
Answer: Partially in Corrosion Analyzer. We do not model the actual full pipeline simulation.
ScaleChem
Which thermodynamic framework is more accurate for ScaleChem calculations?
The answer to this is "Depends." Starting with version 9.6.1 we developed the MSE-SRK thermodynamic framework. This was specifically developed for light hydrocarbons at high pressure. The link to MSE-SRK explains in great detail why this framework was developed.
For most ScaleChem type of calculations OLI recommends MSE-SRK. Remember it is only available in versions 9.6.1 or later.
The option to use the MSE vs AQ thermodynamic frameworks depends on several factors. If you have legacy calculations that date back to the ScaleChem - Standard program (which only supported the AQ thermodynamic framework) then you should probably stay with that framework. Otherwise OLI recommends MSE-SRK over just MSE. Of course there are components in MSE that are not in MSE-SRK (such as heavier organic acids) which would mean that you should use MSE instead but these are not typical ScaleChem components.
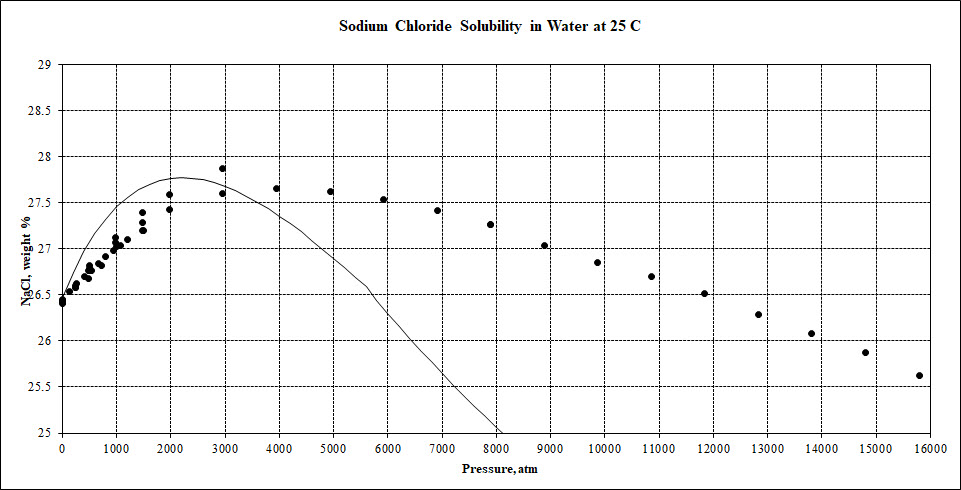
Halite (NaCl) solubility as a function of pressure
It is has been reported that the solubility of halite (NaCl - sodium chloride) has some pressure dependence. Some users have reported a maximum in solubility being reached near 500 bar and then the solubility decreasing. OLI has found data to much high pressures and has the following plot.
The maximum that is predicted is about 2000 atmosphere with the data being predicted slightly higher. This is at 25 oC
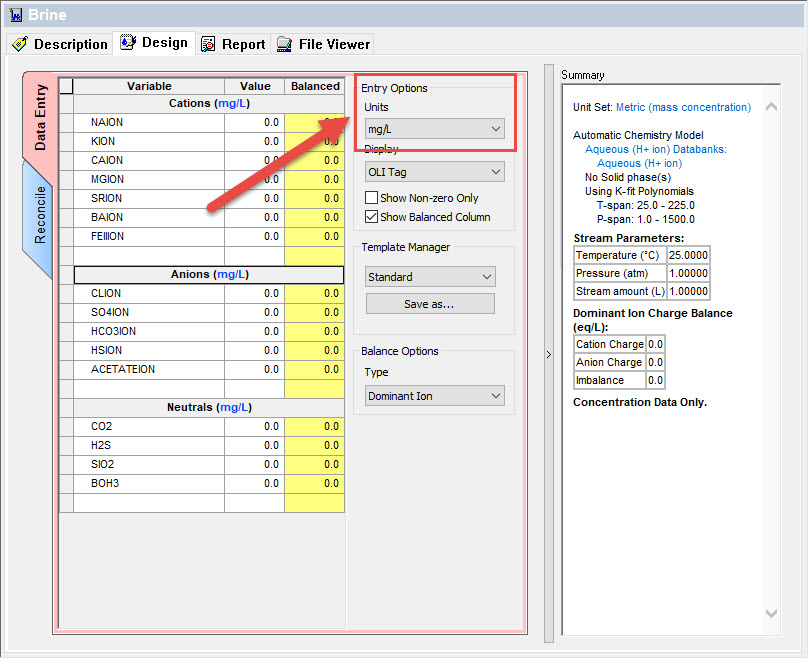
What units can I use for brine entry?
The input units for brine entry are purposely limited to only a select few.
From the brine entry dialog:
Select the Units drop-down box:
You can select from these units. The most common are mg/L and ppm(mass). Sometimes mole % is used as in the following example:
Where did the "Saturate with H2O" button go in Gas and Oil objects?
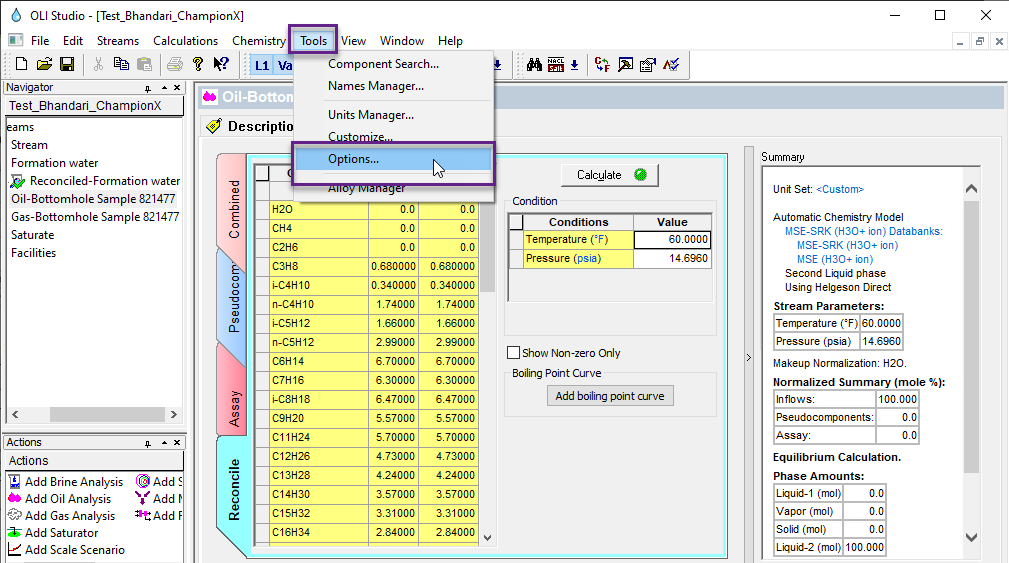
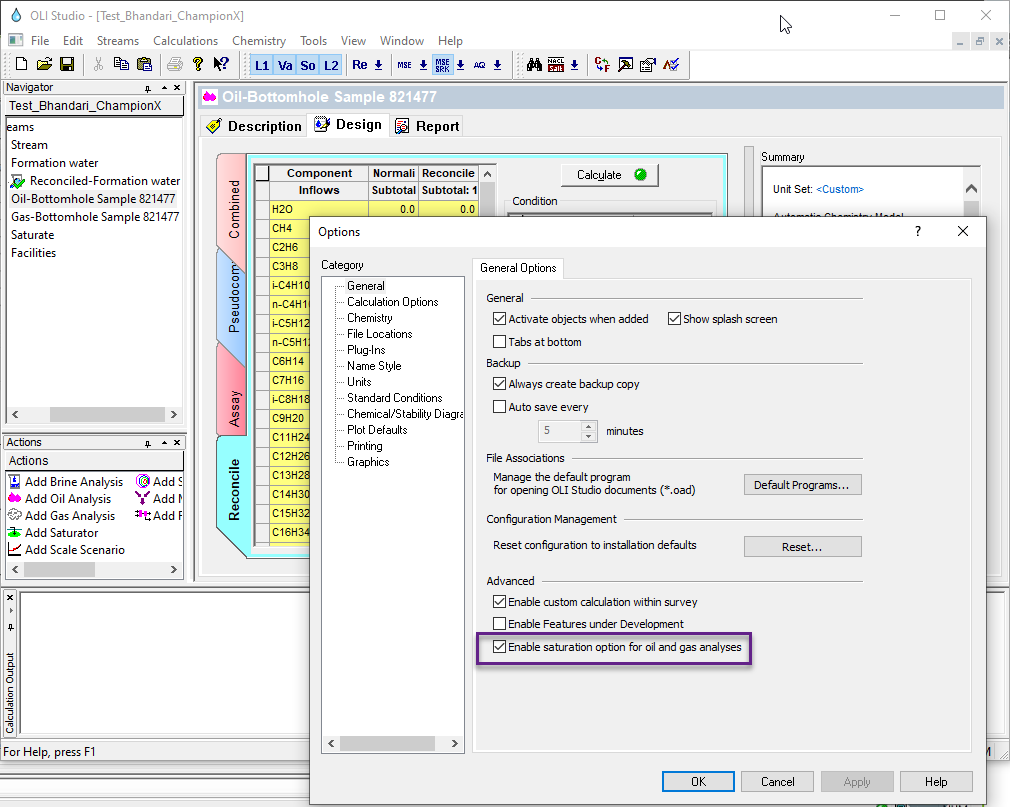
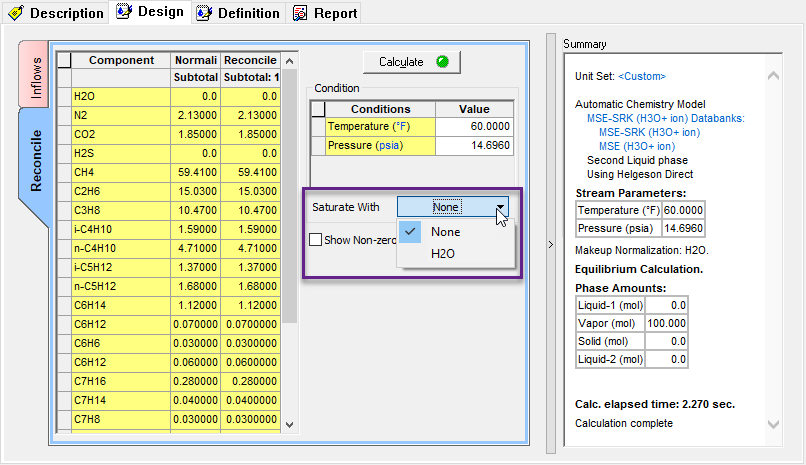
For version 10 of the OLI Studio ScaleChem program we have disabled this feature. It can be tricky to saturate a gas or Oil so it was removed until a more robust calculation can be implemented. However you can enable this calculation if you so require.
From the Tools menu item, select Options...
Locate the "Enable saturation option for oil and gas analysis"
You can now use this option: