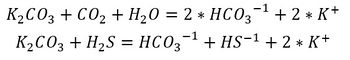Benfield process
Benfield Process
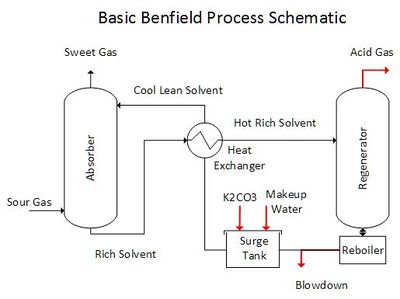
The Benfield Process is a technique used to remove CO2 and H2S from petroleum and industrial gases. The image below is a generalized Benfield Process schematic. The process includes a gas absorption and an alkali regeneration step. Potassium carbonate (K2CO3) is the alkaline absorption solvent. It removes CO2 and H2S from the Sour Gas via acid-base chemical reactions within the Absorber tower. The solvent is then regenerated by evaporating CO2 and H2S using steam or indirect heat within the Regenerator tower.
Chemical Mechanism in the Absorber and Regenerator
The chemical speciation of the two acid gases are well known and reversible,
The reactions proceeds from left to right in the Absorber unit and from right to left in the Regeneration unit.
Steady-state processing
Ideal mass balance is achieved when the CO2 and H2S moles absorbed from the Sour Gas equals the CO2 and H2S moles exiting with the Acid Gas. Deviation from ideal mass balance will occur if CO2, H2S, and also H2O absorption or removal are out of balance. Also, the buildup of impurities entering with the Sour Gas will cause changes in the composition of the alkali solvent. Thus, a blowdown stream is used to maintain consistent solvent properties, and makeup K2CO3 and H2O are added as needed.